Published Oct 27, 2024 | 7:00 AM ⚊ Updated Oct 27, 2024 | 1:11 PM
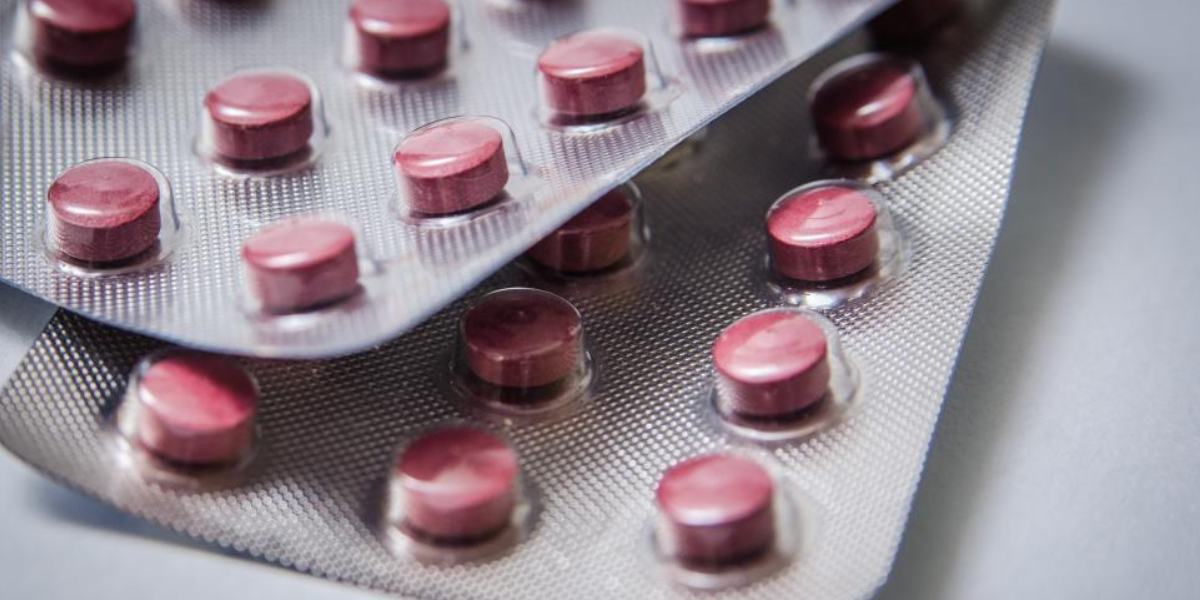
Risks of Look-Alike, Sound-Alike medicines in India, how to prevent medication errors,
Recently, on 20 September, the Delhi High Court barred Sanshiv Health Tech from marketing its product CALIKA-P after finding the company guilty of copying the trade dress and design of Mankind Pharma’s CALDIKIND-P syrup.
The court stressed the importance of protecting consumers, particularly children, from confusion between pharmaceutical products. Justice Saurabh Banerjee presided over the case, in which Mankind Pharma accused Sanshiv of trademark infringement and unfair trade practices, highlighting the misleading similarities in packaging — especially in the colour scheme and design.
This issue goes beyond just the above medication. A wider problem exists in the circulation of many medicines that look or sound alike, contributing to significant confusion among patients.
One major concern in the pharmaceutical industry is the risk posed by “Look-Alike, Sound-Alike” (LASA) medicines. An article published in The Lancet, titled Look-alike, Sound-alike (LASA) Drugs in India, has brought this issue to light, emphasising the dangers these medications present.
LASA drugs frequently lead to medication errors, which can pose serious risks to patient safety.
South First spoke with Dr Parth Sharma, one of the authors of the article, and a community physician and public health researcher. He discussed the dangers posed by identical or deceptively similar brand names of medications, especially in low- and middle-income countries like India, where such errors are particularly prevalent.
Q. What are “Look-Alike, Sound-Alike” (LASA) medicines? Can you explain this concept to someone unfamiliar with pharmaceutical terms?
A: The term LASA stands for “Look-Alike, Sound-Alike” medicines, and as the name suggests, these are drugs that either look or sound similar. This can refer to the way the drug’s name is spelt or how the packaging appears. Let me give you an example from cancer treatments: there’s a drug called Vincristine, and another called Vinblastine. Both start with “Vin-” and end with “-ine.”
Now, there’s an interesting cognitive phenomenon we experience, even as kids, where we can read jumbled words in a sentence because our brains can make sense of them by focusing on the first and last letters. This ability sometimes leads to confusion in drug names as well. When the first and last letters are similar, it’s easy for someone — whether a healthcare provider or patient — to mix them up. So, someone might mistakenly pick up Vinblastine instead of Vincristine due to this similarity. This is an example of a “look-alike” issue.
“Sound-alike” problems arise from brand names. In India, for instance, there’s a drug called Medzol. However, there are at least three different medications with the same brand name, Medzol, but each contains a different active ingredient. If a doctor writes only the brand name without specifying the drug’s active ingredient (what we call the “salt”), the pharmacist may not know exactly which medication the doctor intended.
This can lead to serious issues, as the pharmacist might dispense a completely different drug under the same brand name.
Q. How do packaging similarities contribute to this confusion?
A: Packaging can also lead to confusion. For example, an anti-hypertensive drug and a diabetes medication might be packaged similarly — they may both be round, white tablets and come in blister packs with a brown colour on the back.
If the patient isn’t paying close attention, they could easily confuse one drug for the other. This is particularly problematic in countries like India, where a large portion of the population is semi-literate or struggles with reading in English, which is the language in which most of the medication information is printed.
People often identify their medications based on the tablet’s appearance rather than its name. For instance, a diabetic patient might mistake a blood pressure pill for their diabetes medication simply because both tablets look similar.
This creates a dangerous situation, especially for those who aren’t accustomed to turning the tables around and reading the fine print. We’ve seen many patients who identify their medications by saying things like, “The long tablet is for my cough, and the small round one is for my diabetes.” But if those tablets look alike, the chances of taking the wrong medication increase significantly.
Q. How do similar-looking drugs, particularly in terms of packaging and tablet appearance, impact patients’ ability to correctly identify and use their medications, especially among those with low literacy levels or from rural backgrounds? How does this impact their health?
A: One major issue in our country is that the public health system is overburdened, especially in rural areas, where it caters primarily to low-income populations. Since private healthcare is often unaffordable, people rely heavily on public health services, which leads to overcrowded facilities. Pharmacies are no exception —patients are often in a rush, and impatient due to illness, and they crowd around pharmacists, demanding their medications.
Now, the problem is compounded by the fact that our healthcare system is understaffed. Pharmacists are often overwhelmed, with 30 or 40 people clamouring for their attention at once. In this chaotic environment, it’s easy for mistakes to happen. For example, because two medications might look similar, a pharmacist might accidentally dispense the wrong drug.
This has happened before. I recall a case where a patient with hypertension, who was prescribed Telmisartan (a medication for high blood pressure), was accidentally given Glimepiride (a medication for diabetes) due to the rush and confusion at the pharmacy. Telmisartan lowers blood pressure, while Glimepiride lowers blood sugar. If a person without diabetes takes a drug that lowers blood sugar, it can be life-threatening because their sugar levels could drop dangerously low.
A diabetic patient is often informed about the symptoms of low blood sugar and what to do—like eating some sugar or a piece of candy—but someone without diabetes wouldn’t know these signs. They may not realise what’s happening until it’s too late. This is where the risk lies, especially for populations with low literacy levels or from rural areas, where patients might not be able to read or understand their medications properly.
Q. Who do you believe bears the most responsibility for creating this confusion — the doctors who prescribe the drugs, the pharmacists who dispense them, the manufacturers who produce them, or the regulatory authorities?
A: This is a very complex issue, and it’s difficult to point fingers at just one group. Each level of the system has a role to play in preventing these errors, and solutions exist at every stage.
At the manufacturing level, for instance, there is a technique called tall man lettering. This involves highlighting the differences in the names of similar-sounding drugs. For example, in the case of Vincristine and Vinblastine, the “Chris” in Vincristine and “Blast” in Vinblastine should be printed in capital letters on the packaging to make it easier for healthcare providers and patients to differentiate between the two.
Regulatory authorities also have a critical role. There are laws in place, like trademark rules, which prevent two drugs from having the same or very similar names. However, the enforcement of these laws is lacking. It is the responsibility of the regulatory bodies to ensure that these rules are strictly implemented, and companies that don’t comply should face penalties. This includes monitoring new drugs before their brand names are approved.
Doctors, too, have a responsibility. Guidelines suggest that doctors should write the generic names of medications, not the brand names. For example, instead of writing “Calpol,” they should write “paracetamol.”
This reduces confusion, as brand names like Medzol might refer to different drugs produced by various companies. However, doctors may sometimes prefer a specific brand due to concerns about substandard or counterfeit drugs, which are also prevalent in India. In such cases, they can write the brand name in brackets after the generic name to ensure clarity.
At the pharmacist’s level, pharmacies should store similar-looking or similarly-named drugs on separate shelves to minimise the risk of mix-ups. Pharmacists must be trained to recognise and handle LASA drugs carefully to avoid errors.
In our paper published in The Lancet with co-authors Murali Neelakantan and Ashish Kulkarni, we outlined specific steps that can be taken at every level — from manufacturing to regulation, prescription, and dispensing — to reduce the likelihood of these errors and protect patient safety.
Q. Why are drug manufacturers producing medications with similar names, especially when one is for hypertension and another for diabetes? Is there any benefit for them, or is this due to the regulatory framework in our country?
A: Yes, there is some benefit for manufacturers in using similar names, especially when a brand becomes popular. Famous brand names often become associated with certain conditions. For instance, when someone has a gastric problem, they might immediately think of Pan-D, or when they have a fever, Dolo is often the go-to name. These brands have become household names, so manufacturers sometimes try to capitalise on that by selling their drugs with similar names, hoping to piggyback on the reputation of well-known brands.
However, this also highlights a regulatory failure. Ideally, the regulatory framework should prevent such similar-sounding or similar-looking names from being approved, but enforcement is weak, allowing these issues to persist.
Q. At the policy, manufacturing, healthcare provider, and drug dispensing levels, what do you think are the most urgent actions needed to regulate LASA drug errors? What can be done?
A: In the current scenario, where the policy framework is weak and implementation is slow, immediate action should focus on doctors and pharmacists. In the short term, they are the ones who can actively prevent errors by ensuring they prescribe the right drugs, use generic names, and carefully explain the medication to patients. Doctors should take the time to make sure their patients understand which medications they are getting, and pharmacists need to be vigilant when dispensing drugs.
In the long term, the policy framework needs to be standardised. The government has already taken some steps, like requiring certain drugs to be packaged with blue labels or marking Schedule H drugs (which require a prescription) with a red line. There’s also an app or software being developed to help identify similar drug names. These initiatives are important, but they will take time to implement.
Until those changes are fully realised, the responsibility falls on healthcare providers and pharmacists to ensure patients are getting the correct drugs and understand how to use them.
Q. Recently, the news reported that the Central Drugs Standard Control Organisation (CDSCO) put out a list of non-standard quality (NSQ) and spurious drugs, including commonly used medications like paracetamol and amoxicillin. How do we regulate LASA errors in light of this issue, especially when counterfeit drugs are also a concern? Are LASA errors more common in certain categories of drugs?
A: LASA drugs and counterfeit drugs are two different issues. LASA errors occur when two medications look or sound similar but may still be of good quality — they lead to medication errors, such as wrong prescriptions or incorrect dispensing. Counterfeit drugs, on the other hand, don’t have the correct active ingredient or are fake.
For instance, a cancer drug may be sold as a genuine product, but the vial might contain talcum powder instead of the actual drug.
The news about counterfeit drugs, like the talcum powder incident in Nagpur, highlights the danger of fake drugs, particularly for expensive treatments like cancer medications. For diseases like diabetes or hypertension, the primary concern is that the patient’s condition won’t be controlled.
But with cancer drugs, the stakes are much higher — people spend large sums of money, sometimes lakhs of rupees, only to receive ineffective or fake treatments, which can lead to worse health outcomes and financial ruin.
Q. Coming back to LASA drugs, from a public health perspective, how important is it to have better regulation of drug names and packaging to avoid confusion? Should different names and packaging be required, or can we implement other solutions?
A: The first step in solving this issue is ensuring that there is no overlap in drug names. The regulatory authorities need to enforce stricter rules so that two medications with similar-sounding names can’t exist, especially within the same drug category.
Regarding packaging, another solution could be using colour-coded packaging based on the drug category. For example, all medications for diabetes could come in blue packaging or with a specific colour foil, making it easier to identify. Even if the shape or colour of the tablet itself can’t be changed, the packaging can provide a visual cue. Similarly, antibiotics could be packaged in a different colour.
This would be especially helpful in India’s fragmented and overburdened healthcare system, where a lack of clarity in drug packaging leads to confusion and medication errors.
Q. Given that healthcare expenditure is a significant burden on families in India, how does the problem of LASA drugs contribute to this?
A: This is a huge issue. Every year, about 5.5 crore people in India become impoverished due to healthcare expenses, with 3.8 crore of them falling into poverty solely because of the cost of medications. If these patients are given the wrong drugs due to LASA errors, they end up spending more money without seeing any improvement in their health. Their diseases remain uncontrolled, their productivity decreases, and they sink further into poverty.
This is a serious problem for the country. If a large portion of the population is unhealthy and unproductive, it hampers the nation’s overall growth and development. That’s why urgent regulatory mechanisms are needed to address the issue of LASA drugs and improve patient safety.
Q. You mentioned that LASA drugs are a cause of medication errors, but the drugs themselves can be good for the conditions they are prescribed for. Do we have any data or studies from Indian hospitals to quantify how much LASA drugs contribute to medication errors?
A: Yes, there are a few studies from India, but unfortunately, we don’t have a comprehensive national picture of how prevalent the issue is. Most of the data available are from regional studies, and while they show that the problem of LASA drugs is common, we don’t have enough large-scale monitoring or national-level studies to provide clear statistics.
We’ve mentioned some of these studies in the articles we’ve written, but the lack of continuous monitoring means we’re only now starting to fully understand how serious this issue is. However, I’m optimistic that in the coming years, more independent researchers will take an active role in studying LASA medication errors, and we’ll have more concrete evidence.
Q. Finally, how do you see the future of healthcare evolving to minimise the risks posed by LASA drugs? Could technological solutions, like digital prescriptions, be a viable way forward?
A: Yes, I definitely believe that technological solutions, especially digital prescriptions, could play a major role in reducing LASA-related errors. One of the main advantages of a hospital information management system (HIMS) is that it removes the ambiguity caused by illegible handwriting, which is a common cause of misread drug names. In a digital system, the doctor selects the medication, and the prescription is sent directly to the pharmacist with both the generic and brand names clearly stated. This significantly reduces the chances of errors.
Although packaging and dispensing errors may still occur, digital systems can solve a large part of the problem by streamlining the prescription process. Digital screening methods could also be implemented to prevent companies from choosing brand names that are too similar to existing drugs. The government’s push toward digitalisation in healthcare, like the Ayushman Bharat Digital Mission, is trying to establish hospital information management systems nationwide, and I believe this will be a key step in addressing the LASA problem.
(Edited by Muhammed Fazil)

